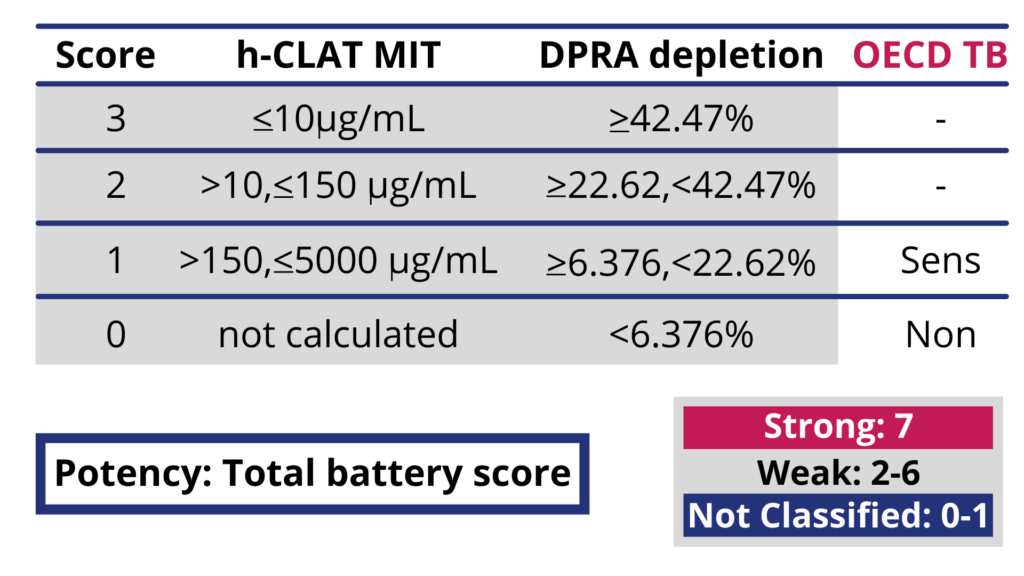
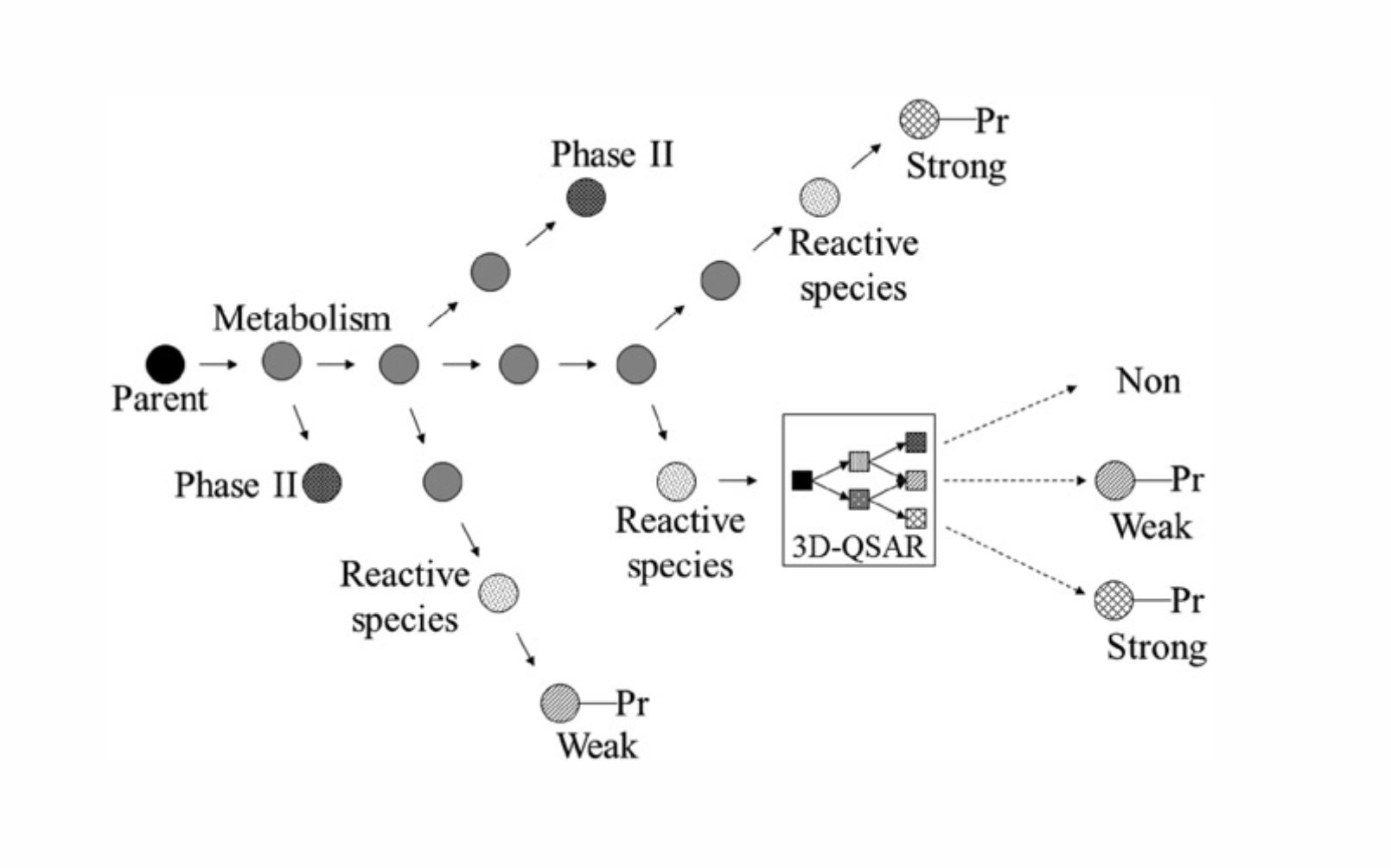
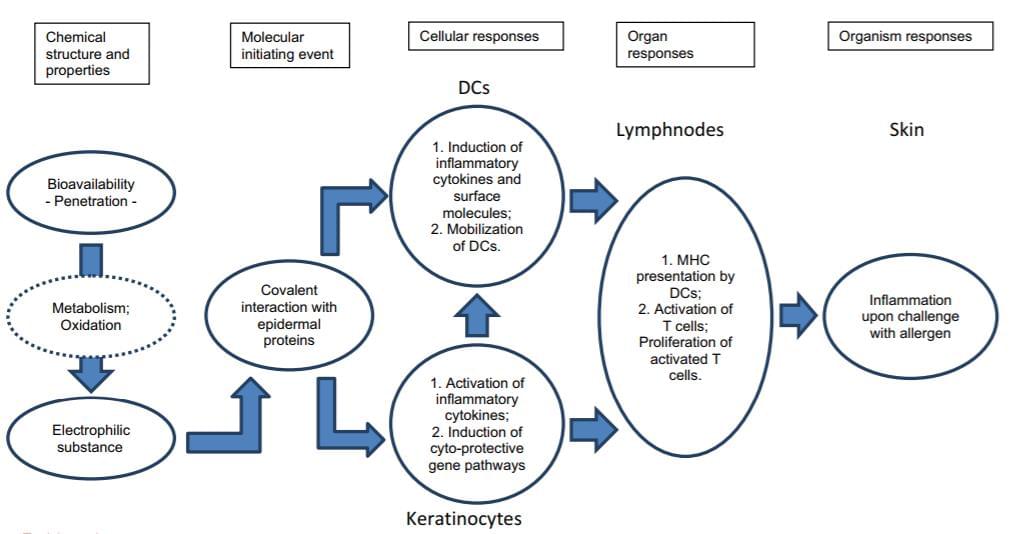
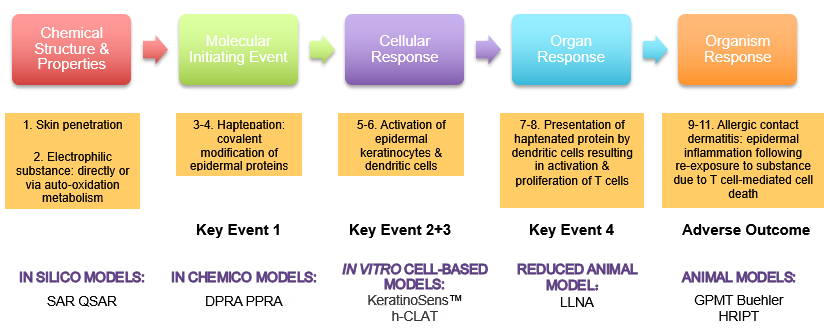
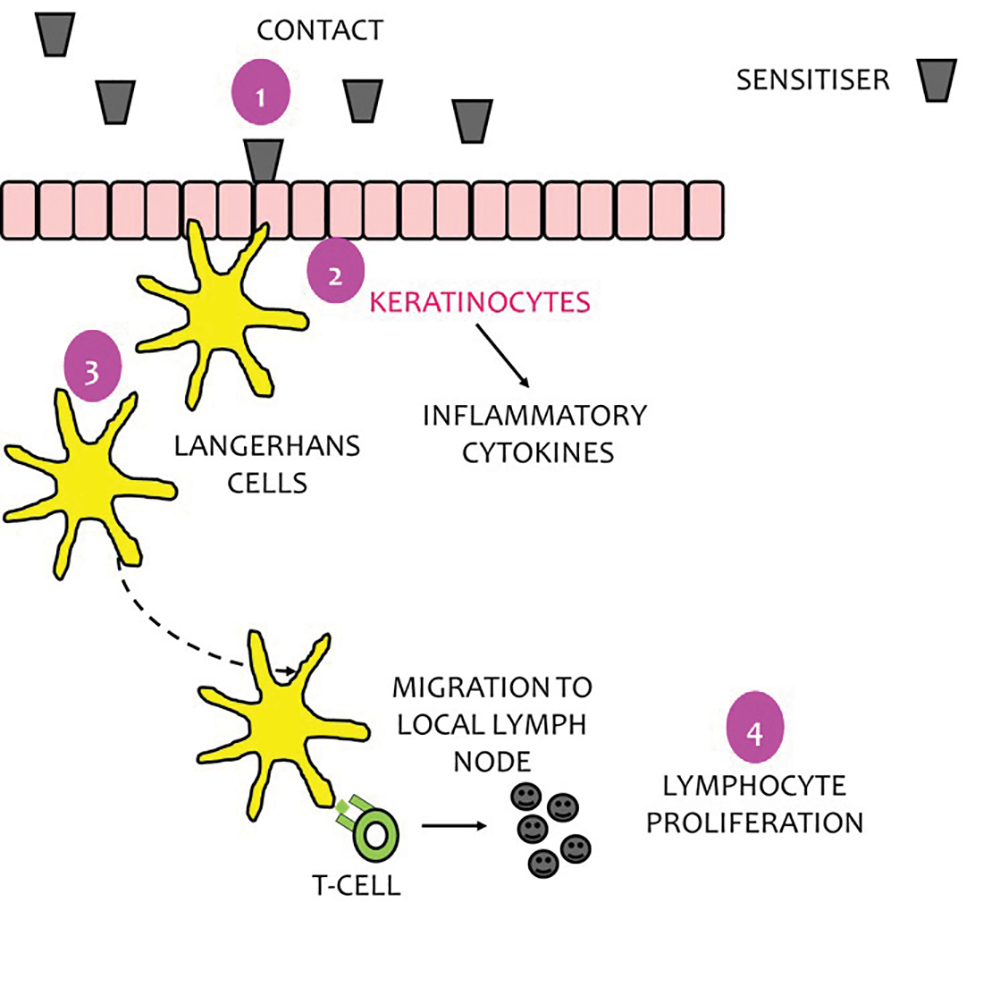
Schematic overview of the mechanisms underpinning skin sensitization... | Download Scientific Diagram

Applying the skin sensitisation adverse outcome pathway (AOP) to quantitative risk assessment - ScienceDirect

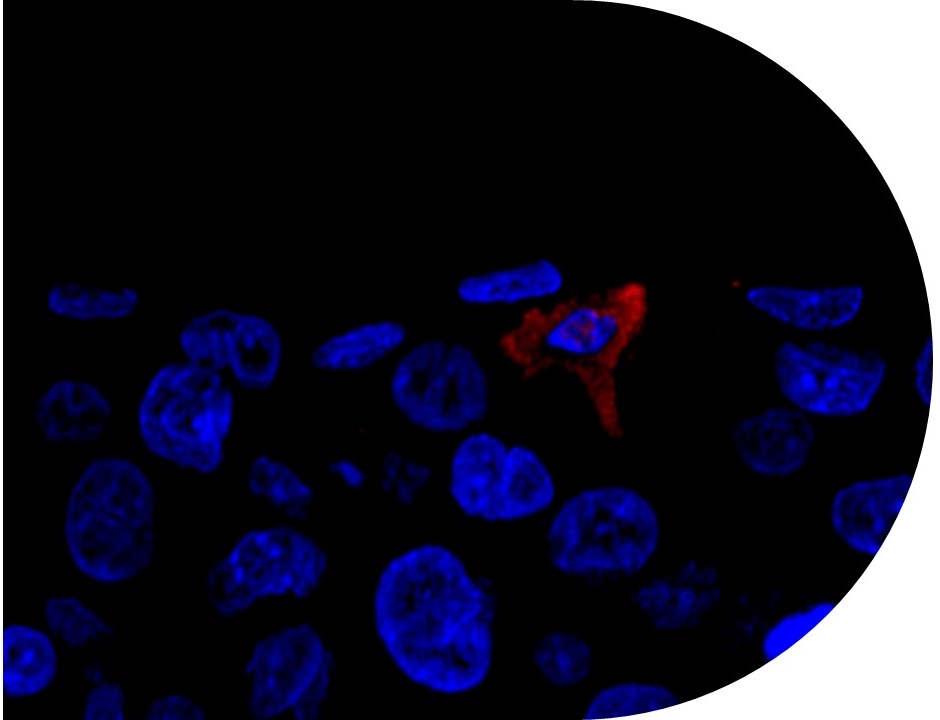
Skin Irritation and Sensitization: Mechanisms and New Approaches for Risk Assessment | Semantic Scholar
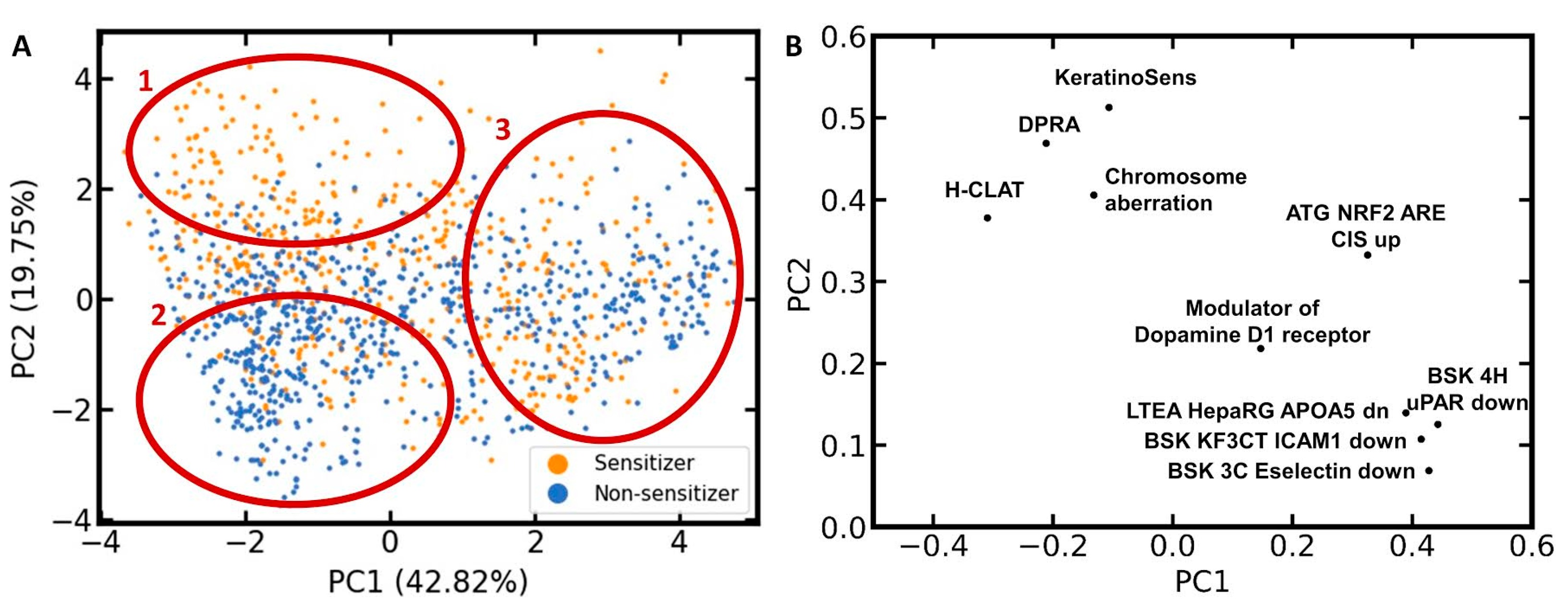
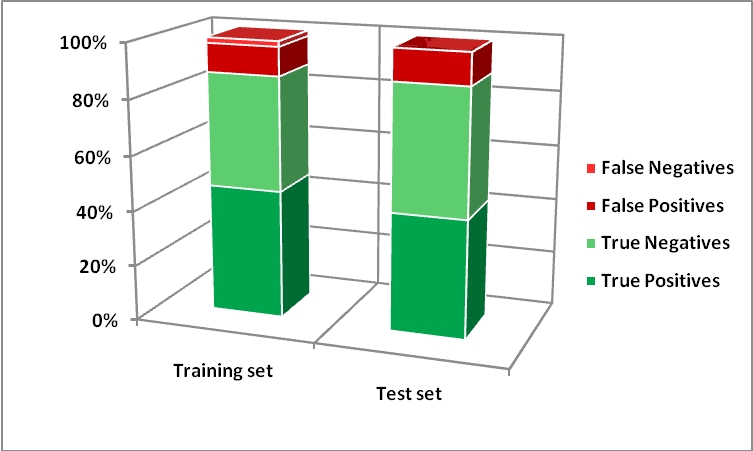
Pharmaceuticals | Free Full-Text | Predicting the Skin Sensitization Potential of Small Molecules with Machine Learning Models Trained on Biologically Meaningful Descriptors

Figure 1 | Skin sensitization testing needs and data uses by US regulatory and research agencies | SpringerLink
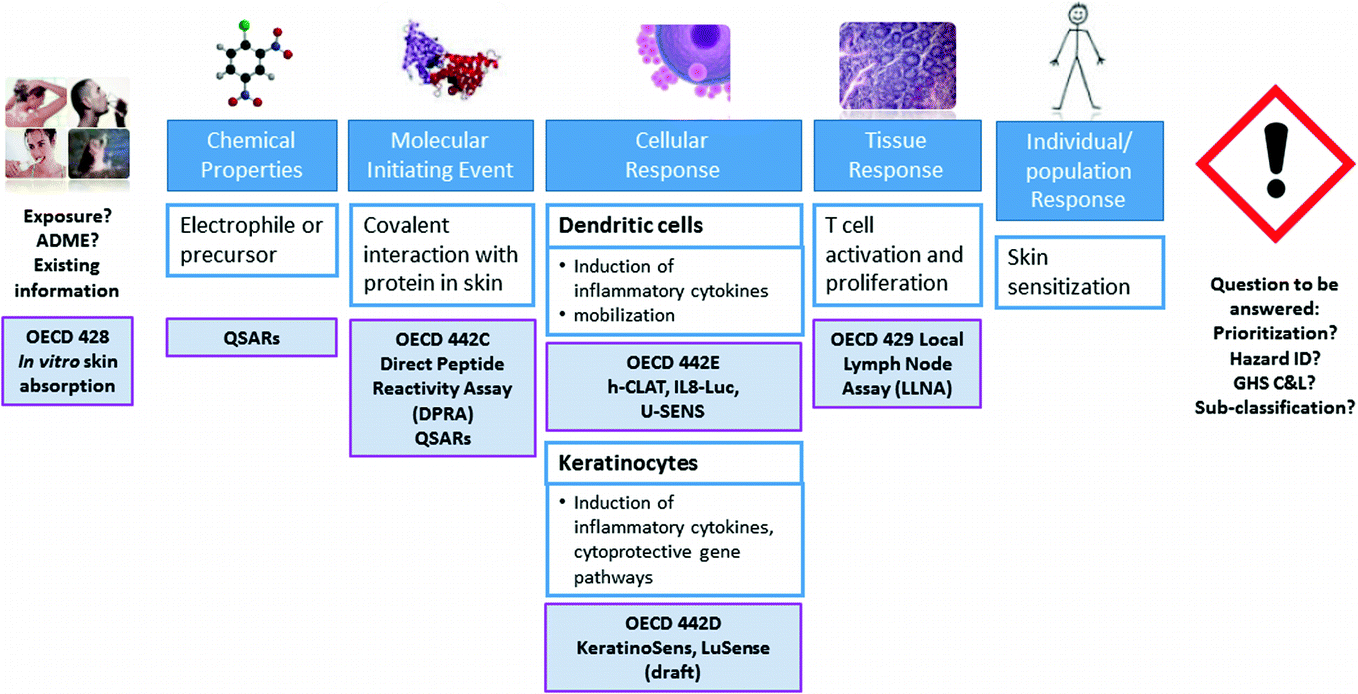
The Use of Adverse Outcome Pathways (AOPs) to Support Chemical Safety Decisions Within the Context of Integrated Approaches to Testing and Assessment (IATA) | SpringerLink