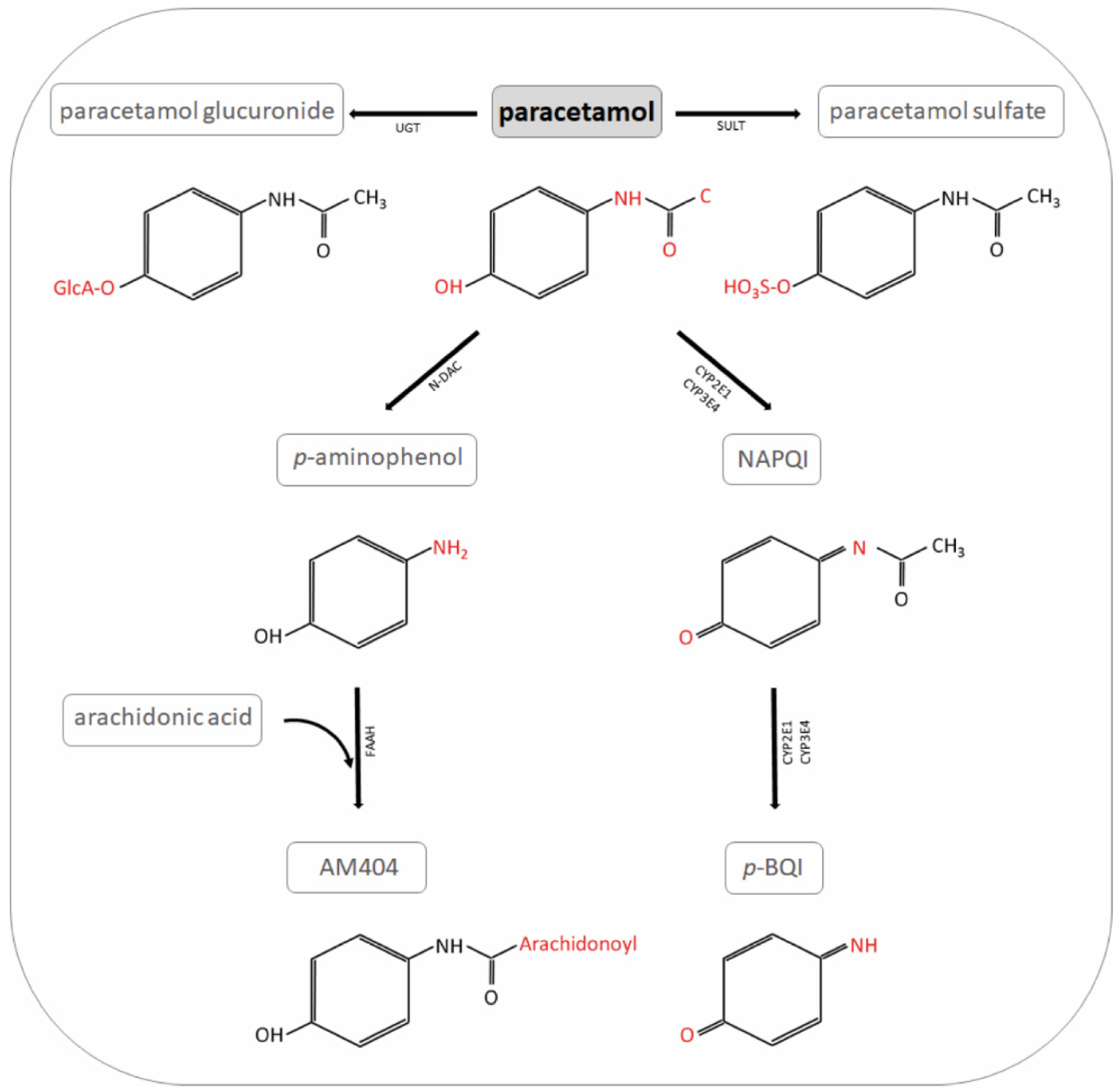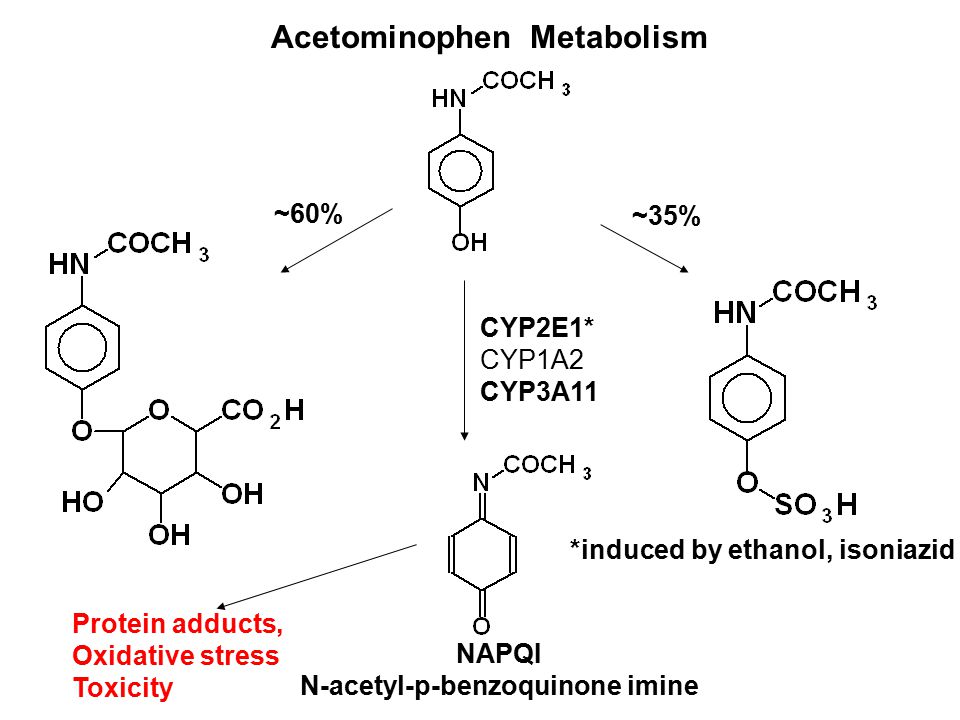
Liver injury induced by paracetamol and challenges associated with intentional and unintentional use

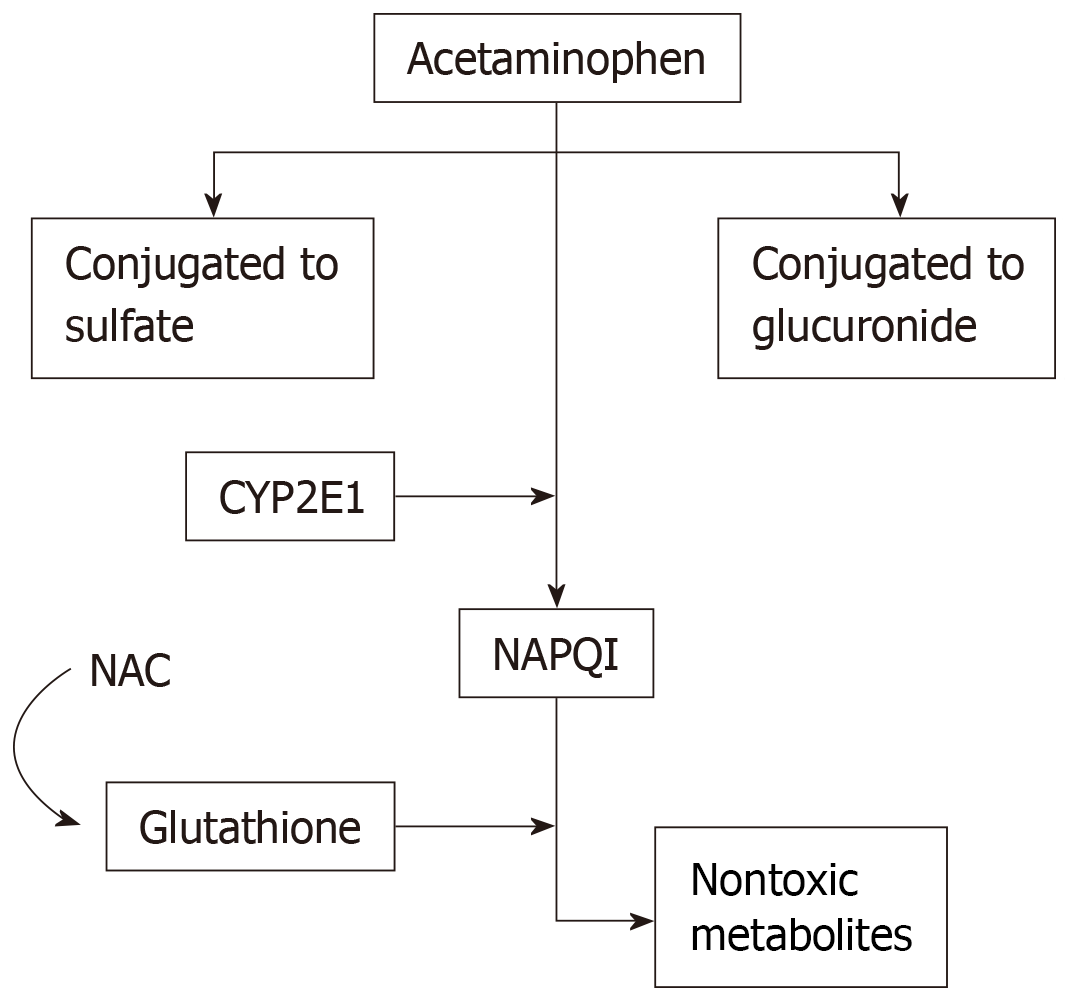
Acetaminophen metabolism. NAPQI, N-acetyl-p-benzoquinone imine; CYP450,... | Download Scientific Diagram
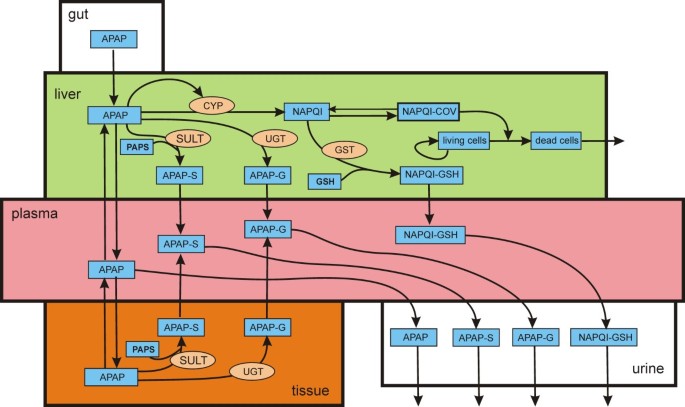
The biochemistry of acetaminophen hepatotoxicity and rescue: a mathematical model | Theoretical Biology and Medical Modelling | Full Text

N-Acetyl-4-benzoquinone imine (NAPQI, N-Acetyl-p-benzoquinonimine, CAS Number: 50700-49-7) | Cayman Chemical

LC-MS analyses of N-acetyl-p-benzoquinone imine-adducts of glutathione, cysteine, N-acetylcysteine, and albumin in a plasma sample: A case study from a patient with a rare acetaminophen-induced acute swelling rash. | Semantic Scholar

Transformation of Acetaminophen by Chlorination Produces the Toxicants 1 , 4 -Benzoquinone and N-Acetyl-p-benzoquinone Imine | Semantic Scholar

The amelioration of N-acetyl-p-benzoquinone imine toxicity by ginsenoside Rg3: the role of Nrf2-mediated detoxification and Mrp1/Mrp3 transports

In-source formation of N-acetyl-p-benzoquinone imine (NAPQI), the putatively toxic acetaminophen (paracetamol) metabolite, after derivatization with pentafluorobenzyl bromide and GC–ECNICI-MS analysis - ScienceDirect

Activation of acetaminophen (paracetamol) to N-acetyl-p-benzoquinone... | Download Scientific Diagram